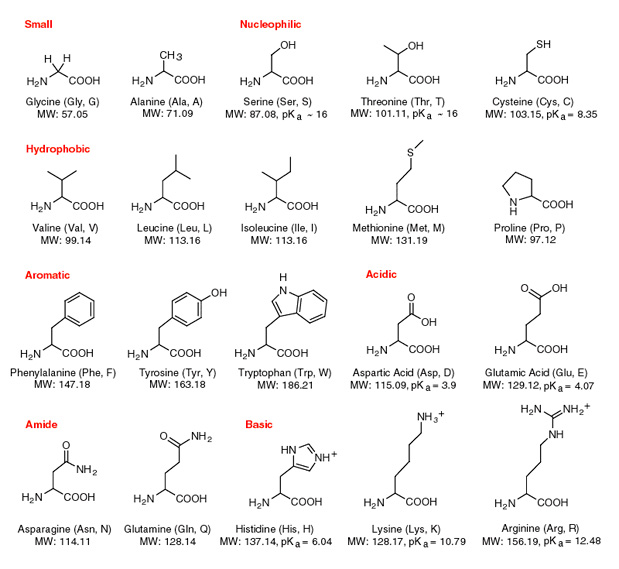
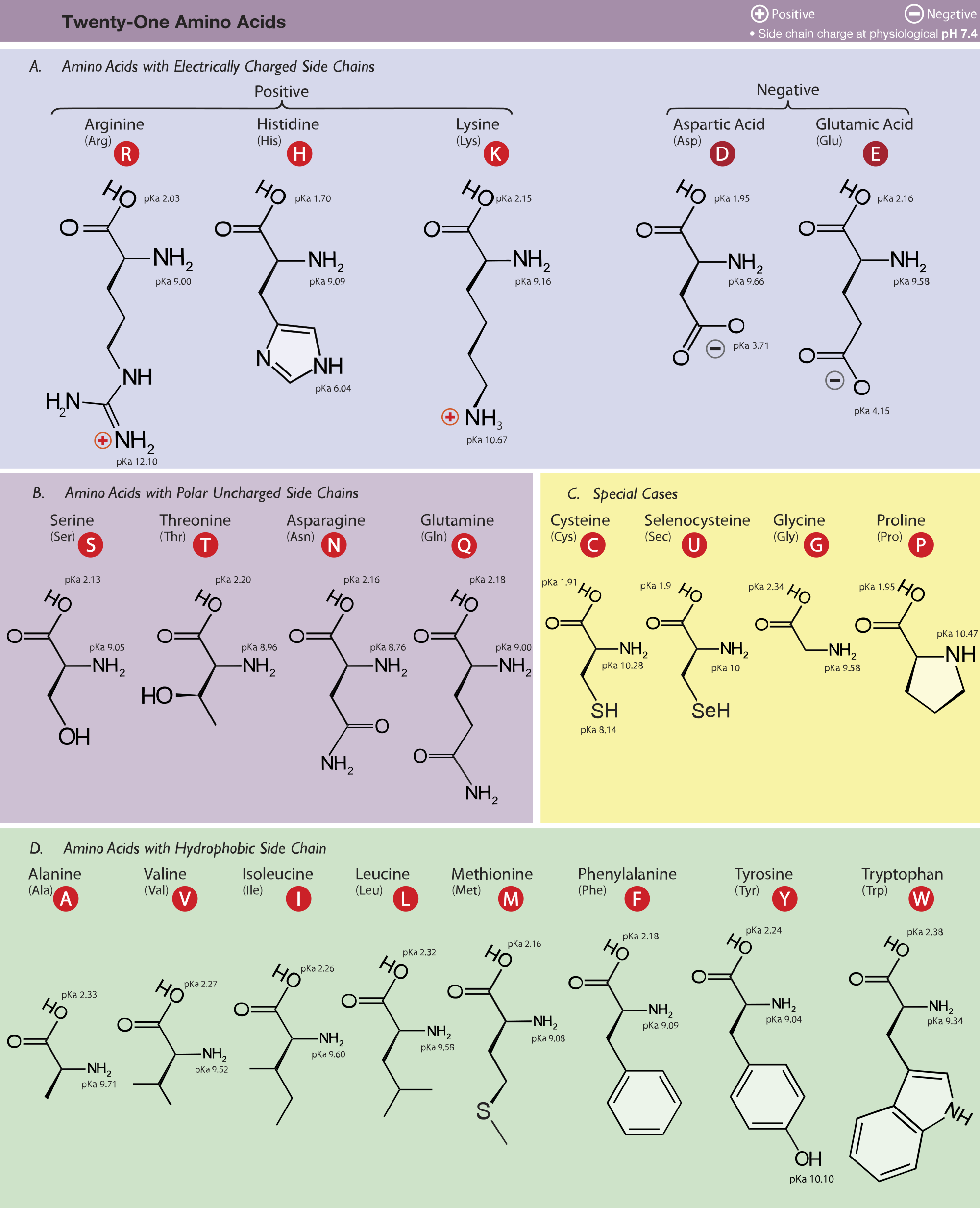
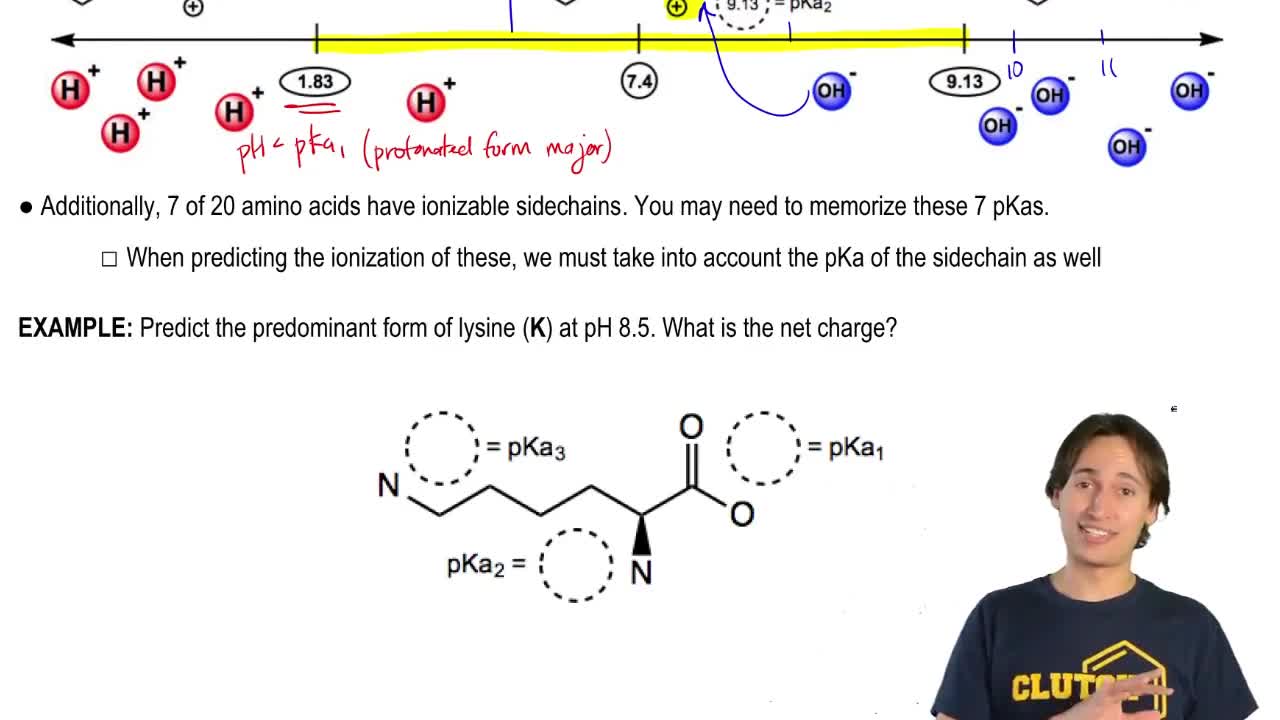
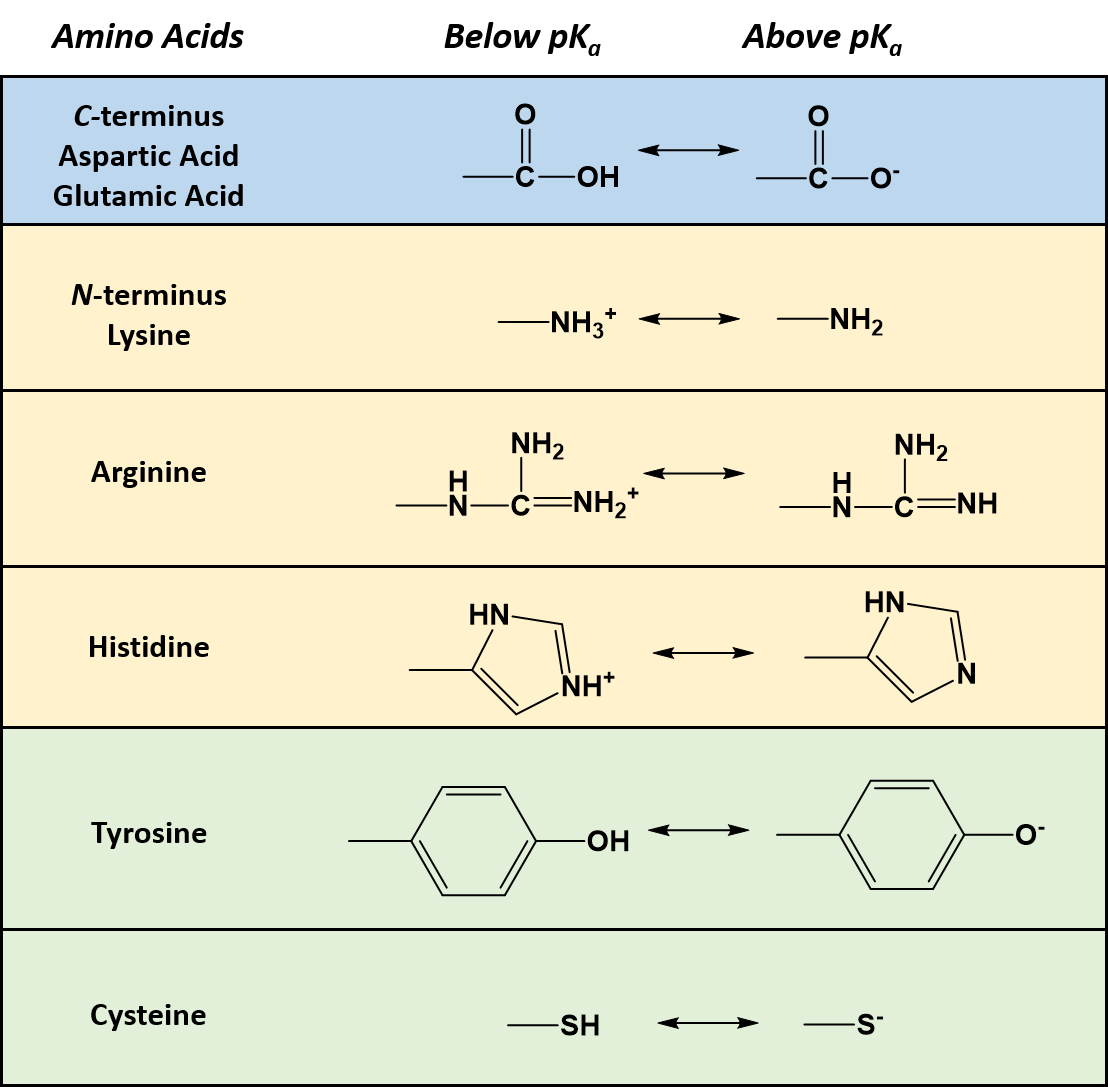
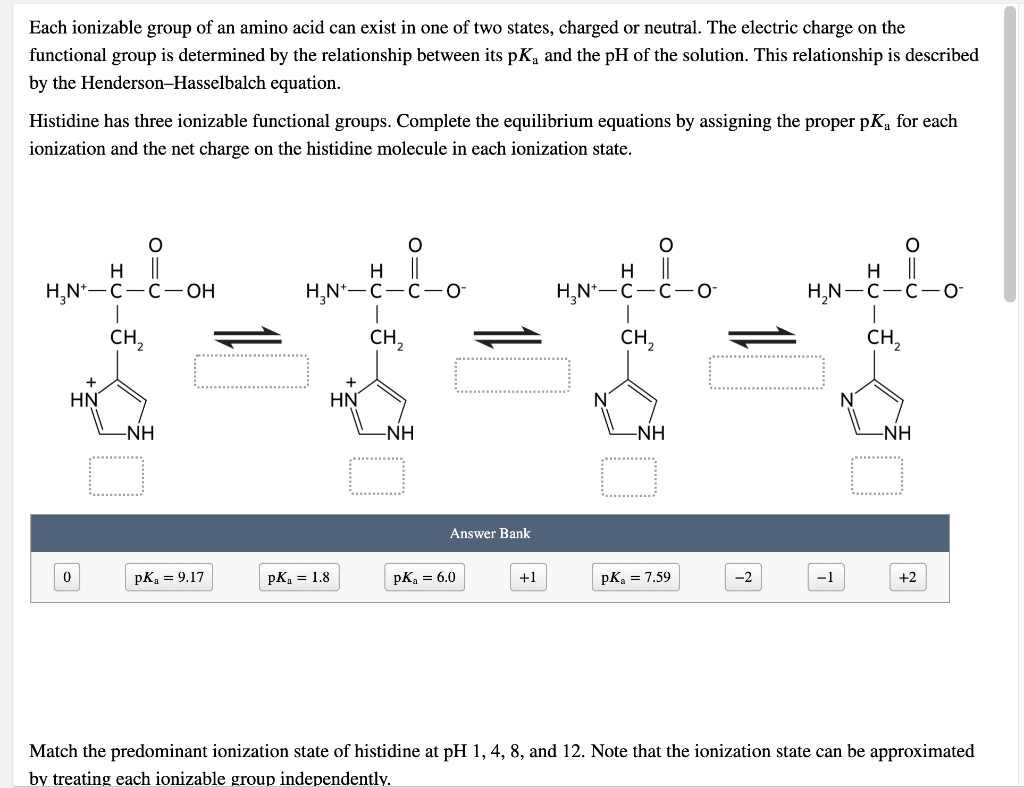
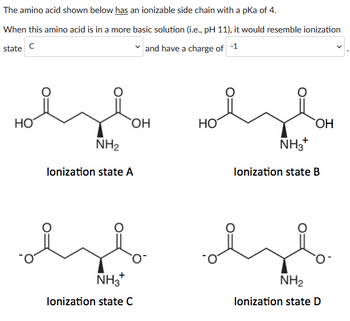
All amino acids have two ionizable groups (an alpha-amino group with pKa = 9.3, and an alpha-carboxyl group with pKa = 2.2). Lysine also has an ionizable side-chain (R) with a pKa
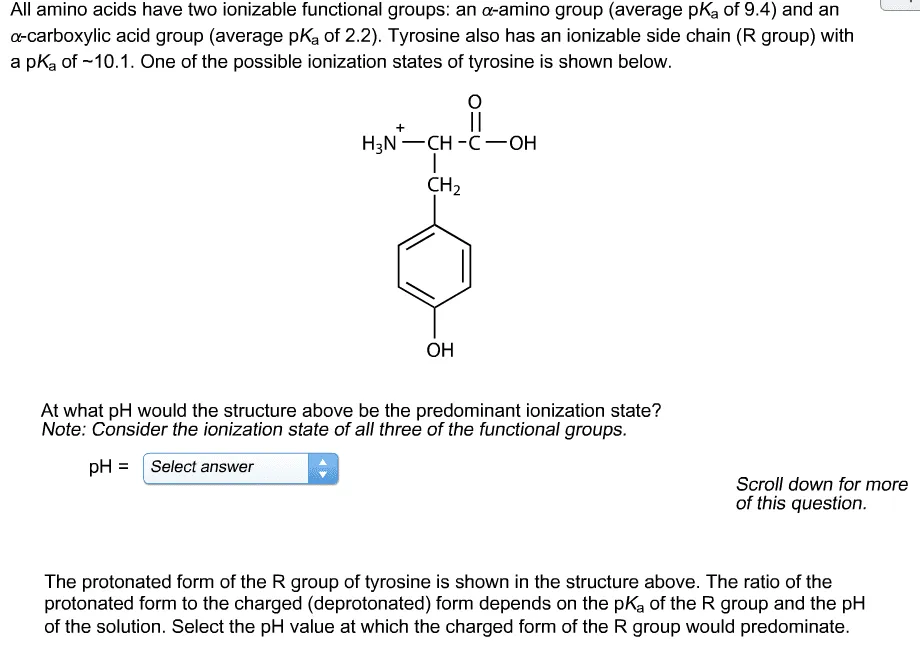
OneClass: All amino acids have two ionizable functional groups: an α-amino group (average pKa of 9.4...

The isoelectric point(pl) of an amino acid is the pH of wihc it has no net charge. The pl of an amino acid that does not have an ionizable side chain -
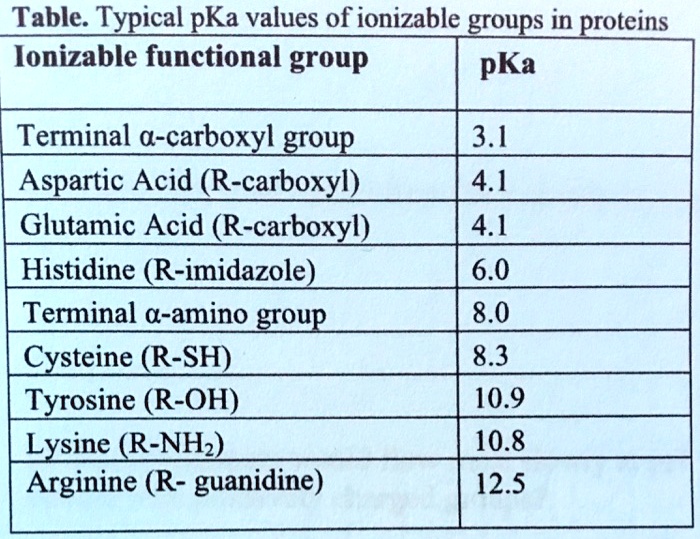
SOLVED: Table: Typical pKa values of ionizable groups in proteins Ionizable functional group pKa Terminal α-carboxyl group Aspartic Acid (R-carboxyl) Glutamic Acid (R-carboxyl) Histidine (R-imidazole) Terminal α-amino group Cysteine (R-SH) Tyrosine ...

Factors influencing the p K values of ionizable groups in proteins. A ,... | Download Scientific Diagram
![The \\[pKa\\] values for the three ionizable groups X, Y and Z of glutamic acid 4.3, 9.7 and 2.2 respectively.The isoelectric point for the amino acid is:\n \n \n \n \n (A)-7.00(B)-3.25(C )- The \\[pKa\\] values for the three ionizable groups X, Y and Z of glutamic acid 4.3, 9.7 and 2.2 respectively.The isoelectric point for the amino acid is:\n \n \n \n \n (A)-7.00(B)-3.25(C )-](https://www.vedantu.com/question-sets/fd71eb03-6e4b-4038-9de0-4e78ab76280c7204254183704236073.png)
The \\[pKa\\] values for the three ionizable groups X, Y and Z of glutamic acid 4.3, 9.7 and 2.2 respectively.The isoelectric point for the amino acid is:\n \n \n \n \n (A)-7.00(B)-3.25(C )-
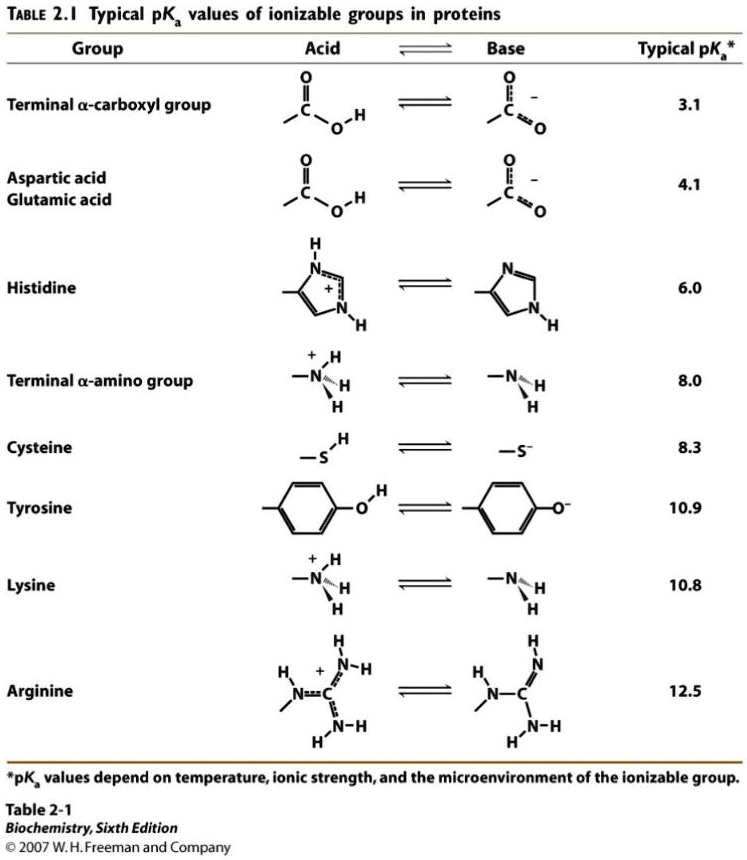
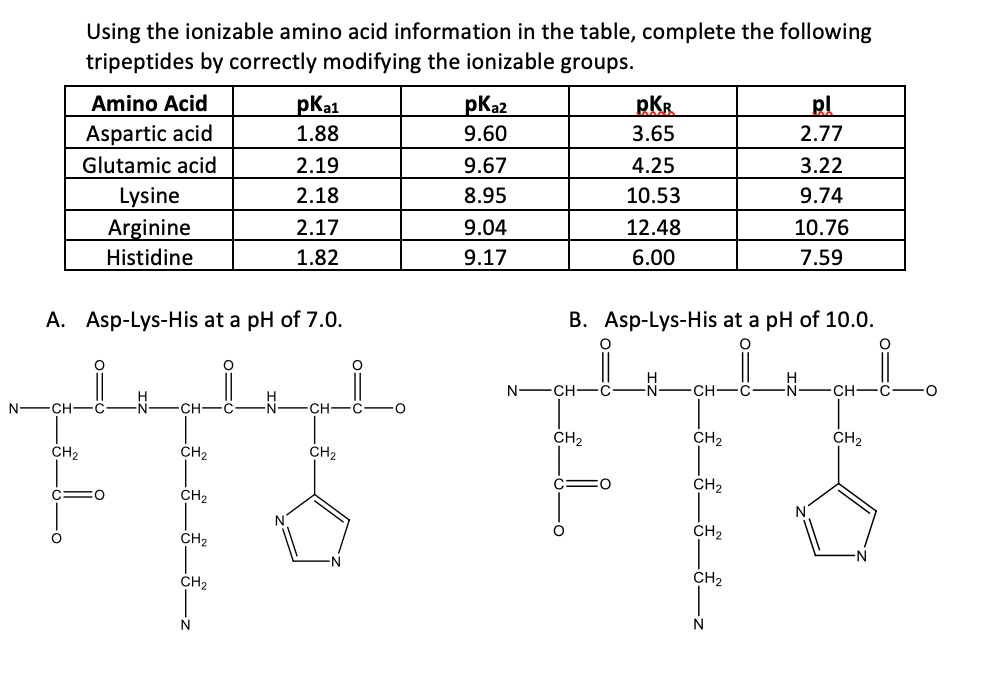
SOLVED: Using the attached pKa table, calculate the isoelectric point of this peptide. TABLE 2.I Typical pKa values of ionizable groups in proteins Group Acid Base Typical pKa* Terminal α-carboxyl group 3.1

Proteins: Amino Acid Chains DNA Polymerase from E. coli Standard amino acid backbone: Carboxylic acid group, amino group, the alpha hydrogen and an R group. - ppt download