Direct amidations between N-Boc protected β-amino acid and C-protected... | Download Scientific Diagram

Resolution of N-protected amino acid esters using whole cells of Candida parapsilosis ATCC 7330 - ScienceDirect

Direct amidations between doubly N-protected α -phthaloyl amino acids... | Download Scientific Diagram

Tertiary-butoxycarbonyl (Boc) – A strategic group for N-protection/deprotection in the synthesis of various natural/unnatural N-unprotected aminoacid cyanomethyl esters - ScienceDirect

Di-Palladium Complexes are Active Catalysts for Mono-N-Protected Amino Acid Accelerated Enantioselective C-H Functionalization | Catalysis | ChemRxiv | Cambridge Open Engage

Halofluorination of N-protected α,β-dehydro-α-amino acid esters—A convenient synthesis of α-fluoro-α-amino acid derivatives - ScienceDirect

A Facile Approach to the Synthesis of Benzothiazoles from N-Protected Amino Acids | Russian Journal of Organic Chemistry
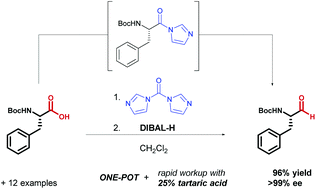
A rapid and efficient one-pot method for the reduction of N-protected α-amino acids to chiral α-amino aldehydes using CDI/DIBAL-H - Organic & Biomolecular Chemistry (RSC Publishing)
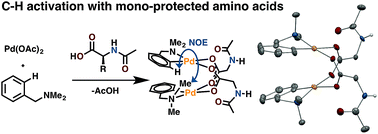
Mono-N-protected amino acid ligands stabilize dimeric palladium(ii) complexes of importance to C–H functionalization - Chemical Science (RSC Publishing)

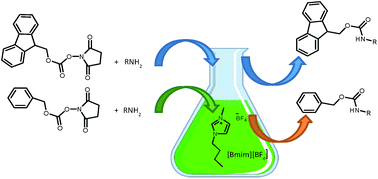
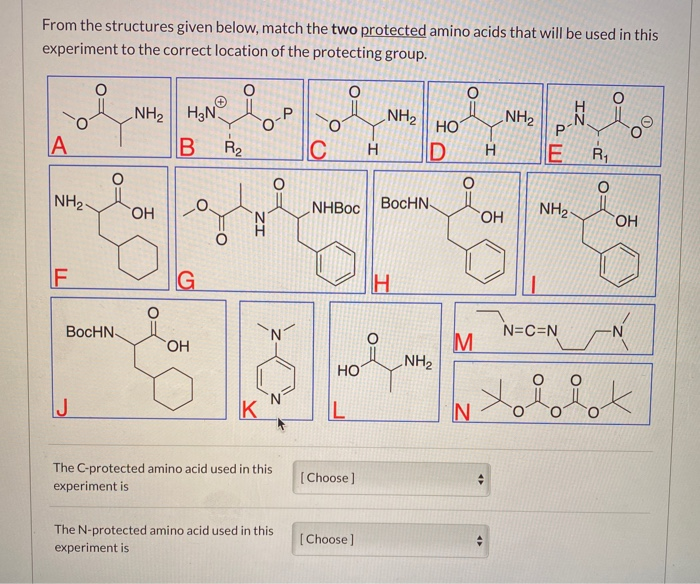

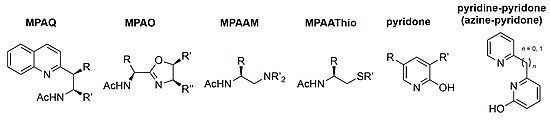

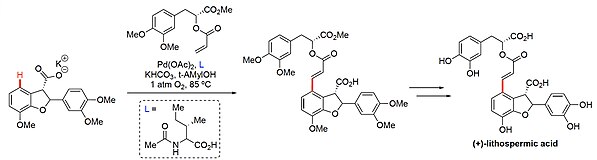

![Boc-Amino Acids [N-Protected Amino Acids] | TCI AMERICA Boc-Amino Acids [N-Protected Amino Acids] | TCI AMERICA](https://www.tcichemicals.com/medias/B1186.jpg?context=bWFzdGVyfHJvb3R8Mjk5MTJ8aW1hZ2UvanBlZ3xoOTIvaGZmLzg5Mjg2NDczODEwMjIvQjExODYuanBnfGU3M2ZlN2FjYmM4ZTQ0ZTNmNmE2MGIwZTFkMDIwMzNkYjA5OTc3MDBjZTgzNGYwOGUzZWU3ZWU0NDcxM2Q0MWI)

![Dnp-Amino Acids [N-Protected Amino Acids] | Tokyo Chemical Industry (India) Pvt. Ltd. Dnp-Amino Acids [N-Protected Amino Acids] | Tokyo Chemical Industry (India) Pvt. Ltd.](https://www.tcichemicals.com/medias/D1031.jpg?context=bWFzdGVyfHJvb3R8Mzg2Njd8aW1hZ2UvanBlZ3xoY2MvaGQ3Lzg5Mjk4MDk2NjE5ODIvRDEwMzEuanBnfGIyYjIzZGVlNjkwNWUwOGZjODM5NDUxZWYwYjE2NDM4YjY5MDYwMzA5NDAwZDQ4NzVkNmRhZGU5N2ZkOTk4MWI)